When a thrombus (blood clot) develops in the cerebral veins and in the dural venous sinuses, the blood flow gets blocked and produces cell damage. This condition is called cerebral venous thrombosis (CVT), although it is also known by other names such as cerebral venous sinus thrombosis (CVST) or cerebral sinovenous thrombosis (CSVT). The continuous pressure that the blood clot creates leads to swelling, which results in very painful headaches. This pressure can ultimately cause the brain blood vessels to burst, resulting in a cerebral hemorrhage. This is a complicated event, as it causes bleeding into the brain tissue, which can kill brain cells.
Cerebral venous thrombosis is a rare type of stroke and it mainly affects children and young adults. According to medical literature, it is estimated that CVT affects 3-4 per million people and 7 per million children. Fifty years ago, CVT reports were based on autopsy findings and it was considered to be a mortal condition. Currently, mortality rates have decreased due to the introduction of neuroimaging and several studies have been carried out in order to determine the prognosis of this disease.
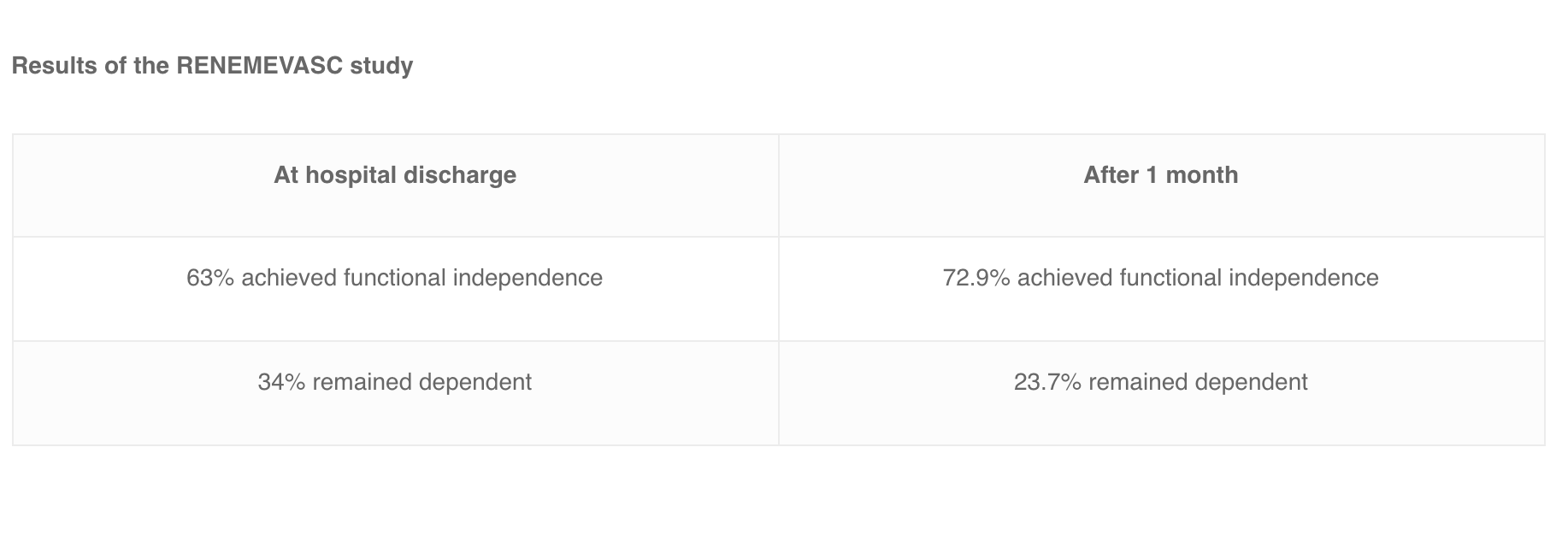
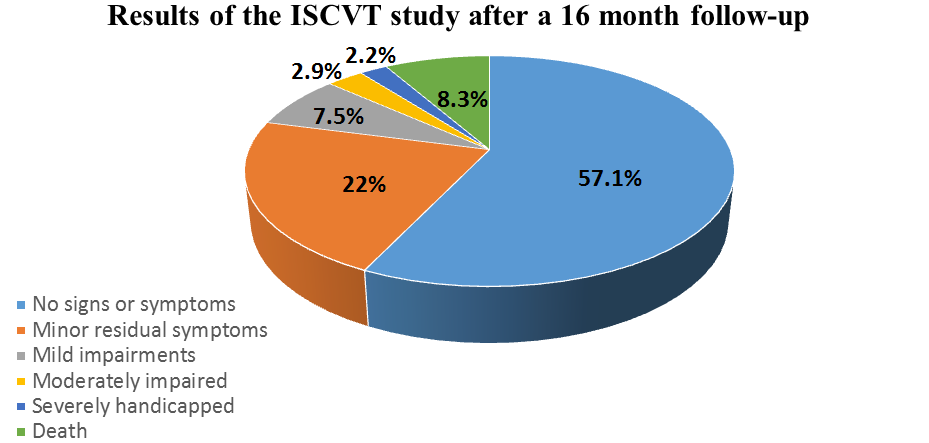
In Mexico, researchers carried out the RENEMEVASC study and found that 63% of the patients achieved functional independence at the point of hospital discharge, whereas 34% of the patients were still dependent. They also noticed some improvements after one month, as 72.9% of the patients achieved functional independence, whereas 23.7% remained dependent. In addition, data showed that recurrence was especially high for patients with pro-thrombotic risks factors or for those who had deep venous thrombosis in lower limbs. The International Study on Cerebral Vein and Dural Sinus Thrombosis (ISCVT) performed the largest prospective observational study of 624 patients with CVT in order to analyze long-term prognosis in several countries. After 16 months, 57.1% of the patients had no signs or symptoms, 22% presented with minor residual symptoms, 7.5% had mild impairments, 2.9% were moderately impaired, 2.2% were severely handicapped and 8.3% had died.[/vc_column_text][/vc_column][/vc_row]
The University of Michigan Health System (UMHS) reports that in approximately 80% of patients, the clots dissolve partially or completely. However, the patients may suffer from headaches and seizures for some time. According to a statement for healthcare professionals from the American Heart Association and the American Stroke Association, 23% of patients present with neurological worsening. This includes depressed consciousness, mental status disturbance, seizures, worsening of or further focal deficit, increase in headache intensity, or visual loss. In the acute phase of the disease, death occurs in approximately 3% to 15% of patients. After the acute phase, deaths are mainly associated with the underlying disorders.
The University of Michigan Health System (UMHS) reports that in approximately 80% of patients, the clots dissolve partially or completely. However, the patients may suffer from headaches and seizures for some time. According to a statement for healthcare professionals from the American Heart Association and the American Stroke Association, 23% of patients present with neurological worsening. This includes depressed consciousness, mental status disturbance, seizures, worsening of or further focal deficit, increase in headache intensity, or visual loss. In the acute phase of the disease, death occurs in approximately 3% to 15% of patients. After the acute phase, deaths are mainly associated with the underlying disorders.
- Fluids
- Antibiotics (in case of an infection)
- Blood thinning medications to prevent the blood from clotting. These anticoagulants are usually injectable (heparin) as a first treatment and then taken orally (warfarin). The American Stroke Association published specific guidelines for treatment in their statement for healthcare professionals in 2011. It indicates that the duration of the treatment with anticoagulants depends on whether the patient has clotting disorders, other risk factors, or if the stroke was of unknown cause
- Thrombolysis therapy
- Antiseizure medicine, in order to control potential seizures
- Intracranial pressure monitoring
- Surgery
- Monitoring of brain activity
- Control of visual acuity
- Try to avoid certain types of medicines, such as oral contraceptives (birth-control pills), as they can increase your risk of blood clots
- Prevent recurrences of CVT with the long-term use of anticoagulants
- Maintain a healthy diet and exercise
- Participate in special rehabilitation or physical therapy, in cases of movement or speech loss





